DWELL TIME
Study title: Nordic Urothelial Cancer Research Group Study on Reduced BCG Dwell-Time in High Risk NMIBC
Short title: NORTH-REG Dwell-Time Study
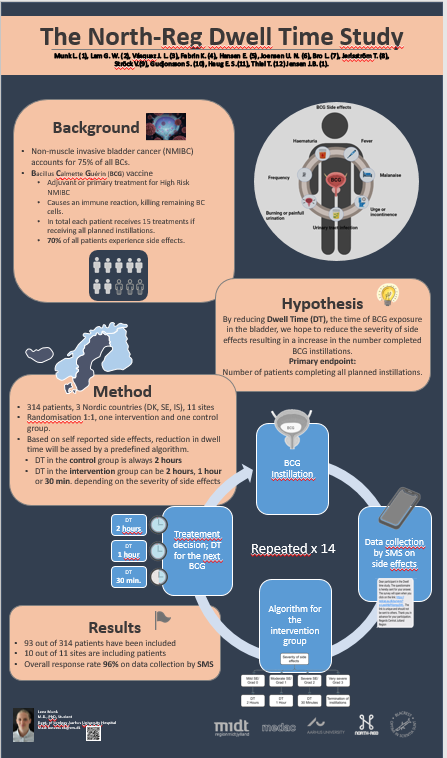
Aim: We aim to investigate whether reducing dwell time (DT), being the time the bladder is exposed to Bacillus Calmette-Guérin vaccine (BCG), in patients experiencing side effects will decrease severity of side effects caused by BCG.
By decreasing BCG side effects we expect to increase the number of patients completing all planned instillations and thereby decrease the risk of recurrence and progression. This resulting in the goal of postponing and potentially reducing the need for cystectomy caused by bladder cancer.
Primary endpoint: Number of Study Subjects completing all 6 induction instillations and subsequent maintenance installations for 12 months (6 + 3 x 3)
Method: This project will include 314 patient through a Nordic collaboration between Denmark, Sweden and Iceland across 11 sites. The patients will be randomized 1:1 into an intervention and a control group. All side effects (SE) will by registered each day during the instillation weeks with daily questionnaires sent on a text message (SMS) to the patient.
Before each instillation, an evaluation is performed, based on the past weeks reported SE. For the first instillation DT is 2 hours regarding both groups. Hereafter, the evaluation will for the patients in the intervention group determine if DT is to be reduced to 1 hour, 30 minutes or continue at 2 hours based on the severity of the SE. Whereas DT in the control group is 2 hours throughout the study. For the purpose of this study, we have developed an algorithm to classify the SE. This algorithm is based on grading of known side effects.
Data collection: We collect data on the patients SE each day during instillation weeks. We sent out text messages (SMS) containing a link to the questionnaire. In this way the data is transferred directly from our database to the patient and back again without the risk of data manipulation.
Location map
Status

Kontakt
Ph.d. studerende
Lene Munk
LENEMU@rm.dk
